CellSpace is a workshop for cellular and molecular sciartists. For our 2020 project, we are creating work for “New Ways of Living.” The artists are located around the world, and are working in multiple media, including traditional painting, digital art, animation, virtual reality, sculpture, and sonification.
Arnthor Ævarsson
Ph.D. Molecular Biophysics
MA Student in Fine Art, Iceland University of the Arts
The Molecular Kiss of Death
At its core, the onset of COVID-19 is a connection between a human molecule and a molecule of the coronavirus. The image shows the binding of the Spike protein on the surface of the coronavirus to a receptor protein (ACE2) on the surface of a human cell. The image is a close-up atomic view of this connection showing only limited parts of these proteins, the human receptor protein in blue and the coronavirus Spike protein in red. The image is based on the structure of the complex between these proteins determined by x-ray crystallography (Lan et al). The atomic coordinates are used to generate the molecular part of the image through molecular graphics (Goodsell et al). One may see the background as a battlefield with the coronavirus against us humans in the form of our response to this biohazard, as indicated by its symbol. The players are on the field in an ongoing battle, but the progress and outcome of the battle remains uncertain. This limited but very specific molecular interaction seen in the image is the determinant of the onset of the disease that eventually may lead to your death. This is the molecular kiss of death.
License: Please contact Arnthor for information on usage and license.

A Note from the Future
Being caught up in the now can mean you do not have a clear perspective of the now, it will only become clear in retrospect. This is a tear-out from a future comic book looking back to the year 2020 in some kind of historic context that is hidden since this is the only piece we see. However, it bears the message that 2020 and the pandemic will mark a profound change in our systems and values towards a more sustainable and humane world, truly the beginning of a new era, a new world order.
License: Please contact Arnthor for information on usage and license.
Reference: https://en.wikipedia.org/wiki/New_world_order_(politics)
Darshak Bhatt
Molecular landscape of Coronavirus
The present scene features a single coronavirus particle in the pulmonary tissue surrounded by respiratory-mucus, antibodies, and various other small proteins with innate immune functions. Each individual molecular component of the scene is shown separately and labeled to make the scene more comprehensive. The virus is composed of various viral proteins and a RNA genome. The spike protein is incorporated in the viral membrane and regulates the attachment and entry into host cells. Along with the spike protein, the envelope protein is also found on the membrane, and it is involved in the budding stage of viral replication cycle. The membrane protein plays an important role in organizing the nucleoprotein inside. While the nucleoprotein is composed of unitary nucleocapsid proteins that help fold and protect the genomic RNA. In the periphery of the virus particle, we can visualize interferons that are anti-viral proteins and function via blocking viral replication inside host-cells. We can also find some defensin and cathelicidin proteins that are known to exhibit antimicrobial activity and regulate immune responses.
SARS-COV2 in a respiratory aerosol
The present scene depicts a possible respiratory aerosol (droplet smaller than 5 µm) containing SARS-COV2 virus particles. Transmission of respiratory infection, can occur through spread of virus-containing droplets exhaled from infected individuals during various activities like breathing, speaking, coughing, sneezing and singing. A large proportion of the spread of coronavirus disease 2019 (COVID-19) appears to be occurring through airborne transmission of aerosols produced by asymptomatic individuals. Therefore, a conventional yet simple and cost-effective control measure to reduce transmission via droplets could be achieved via wearing masks.
Note: This illustration is adapted from the molecular landscape of a respiratory droplet by D.S.Goodsell.
License: This content is published under a CC-BY 3.0 license. You’re free to use this as long as you credit the author ‘Darshak Bhatt’.
To read more: https://science.sciencemag.org/content/368/6498/1422.full
Dr. Disha Chauhan
The Visual Stories Studio
Alternate Doorway: Neuroinvasion by SARS-CoV-2
The illustration shows how binding of SARS-CoV-2’s spike protein to host angiotensin converting enzyme 2 (ACE2) triggers viral entry into epithelial cell, but neuropilin-1 receptor (NRP-1) is an alternative doorway for SARS-CoV-2 to enter and infect cells. In cells with both the ACE2 and NRP-1, SARS-CoV-2 infection is more compared to cells with either ‘doorway’ alone.
License: This media is provided for free use under a CC-BY-4.0 license. For use of this image. please acknowledge “Dr. Disha Chauhan”.
For more information contact:
Email: thevisualstoriesstudio@gmail.com
Twitter: www.twitter.com/Visualscitories

Timothy Davison & Hamed Kaddoura
Developing an iPad application for molecular biology education
Introduction to Translation
Viruses such as SARS-CoV-2 invade our cells and appropriate the cellular machinery used in healthy translation to produce their proteins and replicate themselves. The first step in adapting to new ways of living with SARS-CoV-2 is understanding how it operates within our cells.
In this work, we present an interactive computer simulation with narrative elements that serves as an introductory lesson on the process of translation in a human cell. This application runs on modern Apple iPads and is currently in the prototyping phase. The artistic works of David Goodsell heavily inspire our visual design. We employ a cross-sectioned view of an active, living cell with vibrant colour schemes and scientifically accurate structural biology models using data from the Protein Data Bank (PDB). Users learn fundamental biology by reading the narrative while interacting with the narrative simulation in real-time through a variety of guided and unguided activities. This short video provides a walkthrough of a user’s experience with the narrative simulation. Currently, the subject matter is limited to normal translation within the cell. In the coming weeks, we will be working to extend this prototype lesson to include the introduction of SARS-CoV-2 to show how it uses our cellular processes against us.
To learn more or to follow the progress of this project: @timd_ca
We owe many thanks to David Goodsell and Arthur Olson for their consultation and help with the writing on this project.
License: These images/videos are free to use under the CC-BY-4.0 license. For the use of this media, please acknowledge “Timothy Davison”.
Click here to watch a full length video of the lesson.
Mp4 files are available for the Short Video and the Full Length Lesson.
Sabela DeScience
science illustrator & post-doc at Amsterdam UMC (https://www.sabeladescience.com/)
Neutralising antibodies against Sars-CoV-2
Our body can produce natural defense molecules against the coronavirus – the so-called antibodies. One of the ways antibodies protect us is by neutralizing the virus. Sars-CoV-2 virus is decorated with many spike proteins (in magenta). These are large trimers that protrude out of the viral membrane in different angles; around 24 spike proteins per virion give the coronavirus its peculiar silhouette. The spike proteins of the virus bind the ACE2 protein (turquoise) of the host cell (blue), triggering invasion and infection. When neutralising antibodies (in green) enter into action, they bind and block the spike protein. The virus is then unable to infect cells.

Gloria Fuentes
Since very early in the outbreak, scientific efforts have been essential in providing valuable information on SARS-CoV-2, the agent causing COVID19. Laypeople have become very familiar with the structure of the virus, largely thanks to the impressive images of SARS-CoV-2 rendered by scientific illustrators, where the agent has been shown in all its splendor.
Yet the information is highly dynamic, as the virus is, and is continuously subjected to changes as more studies are conducted.
This animation incorporates data on a particular variant of the virus, D614G, together with structural and morphological characteristics distilled from experiments on intact virions.
The intention has been to show the dynamic properties of the virus, as well as depicting it in a less appealing manner. In spite of this, the magnificent of these cellular machineries has taken its own course and has highlighted the elegance inherent in its biology. Probably in a few months, we will have new data that will change the story behind this animation. This is the way science works, and will always do.
I would like to express my gratitude to all those scientists working against the clock to disclose details on this fascinating virus. Every little piece of information will contribute to the understanding of this agent, and will have an impact on the design of vaccines and therapeutics against SARS-CoV-2.
Molly N. Gilmore
Bioinformatics Software Engineer and Adjunct Instructor, WVU Reed College of Media

Zoonotic Shift: An Age-Old Phenomenon Affected By Current Human Activity
Why are some viruses able to infect both humans and animals?
Recent studies are stating with increasing confidence that the SARS-CoV-2 has a zoonotic origin, most likely from bats. This illustration explores the origins of the virus and the possible roles of human behavior and habitat destruction that increase the likelihood of animal to human transmission of disease.
License: This work is freely available with a CC-BY-4.0 license. For use of this image, please acknowledge “Molly N. Gilmore”.
Sarah Iannucci
MSc Student in Medical Visualisation and Human Anatomy, The University of Glasgow and The Glasgow School of Art

Deactivation
We are all told that good hand-washing habits can help to stop the spread of COVID-19, but how exactly does this work?
“Deactivation” shows the process by which soap surfactants interact with the viral envelope of SARS-CoV-2, tearing apart the structure and rendering the virus inactive. As we scrub our hands with soap, these surfactants are brought into contact with the viral membrane. Soap surfactants have a hydrophilic (or water-loving) head and a hydrophobic (or water-hating) tail. The viral membrane has this configuration too, with the hydrophilic heads facing outward and the hydrophobic tails facing inward; this is the basis for plasma membrane formation. As the surfactant tails encounter the hydrophobic core of the viral membrane, they begin to insert themselves, compromising the viral membrane integrity and leading to the formation of micelles. The surfactants are also attracted to other proteins within the membrane, such as the characteristic spike proteins. Eventually, the membrane is torn apart, the proteins disperse, and the viral contents are exposed. Thus, the simple action of washing our hands regularly and efficiently can effectively render the SARS-CoV-2 virion inactive by this process.
For more information, you can reach Sarah at sarahmiannucci@gmail.com.
License: This work is freely available with a CC-BY-4.0 license. For use of this image, please acknowledge “Sarah Iannucci”.
Sara Jager
Postdoc at King’s College London, UK
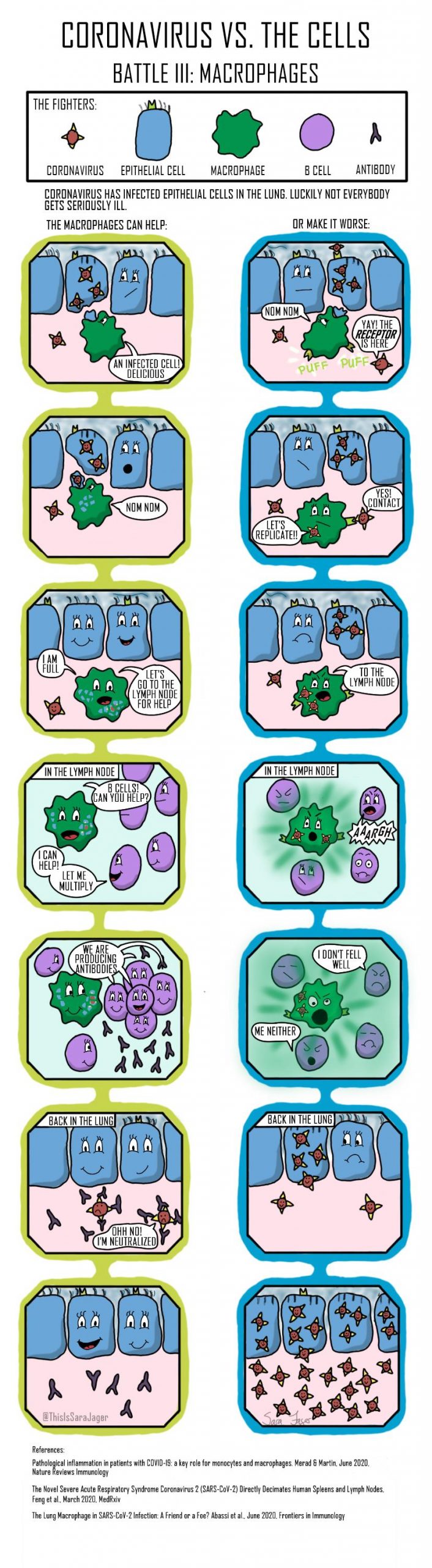
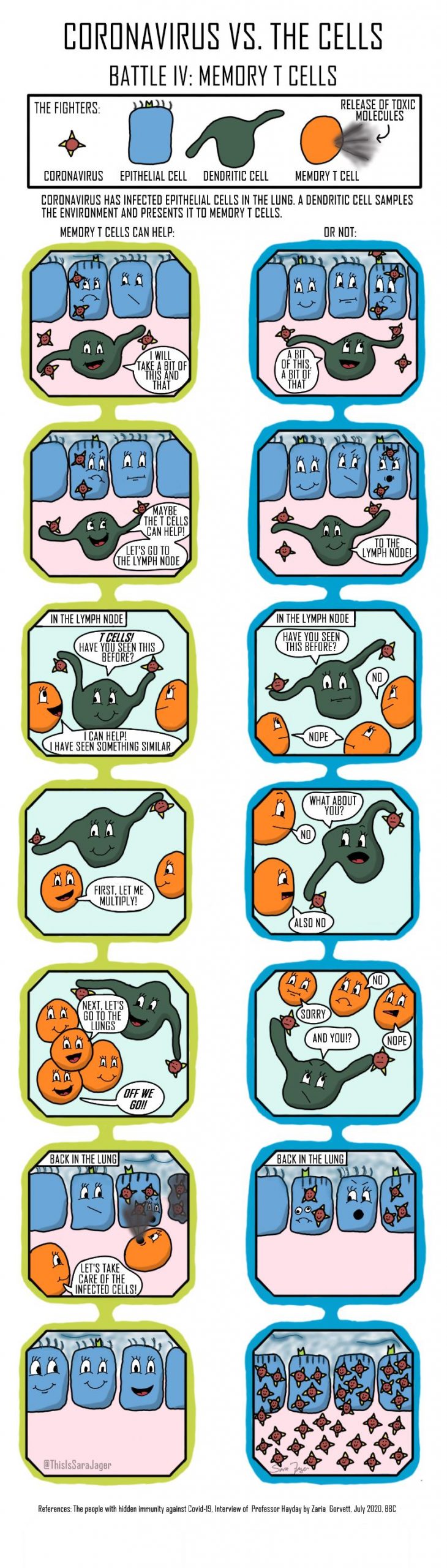
Four Battles
Question: Why do some people get seriously ill with coronavirus while others do not?
The answer to the question is not fully understood and it is unlikely that one answer will explain all the serious cases. I wanted to explore different possible answers on a cellular level and the result is this small comic series. The comics look at four different parts of the immune system’s response to coronavirus.
- At the point of entry, cells in the lung try to repel coronavirus
- After the initial infection natural killer cells can be recruited. The natural killer cells are less responsive in the elderly.
- A deficit of immune cells have been detected in coronavirus patients. Some researchers have suggested that this is due to macrophages becoming infected and behaving inappropriately.
- Previous virus infections can have primed the immune system to handle a coronavirus infection more efficiently resulting in a less severe illness.
Contact: sarajager@msn.com, @ThisIsSaraJager (Twitter)
License: This work is freely available with a CC-BY-4.0 license. For use of this image, please acknowledge “Sara Jager”.
click on these tabs to see the battles:
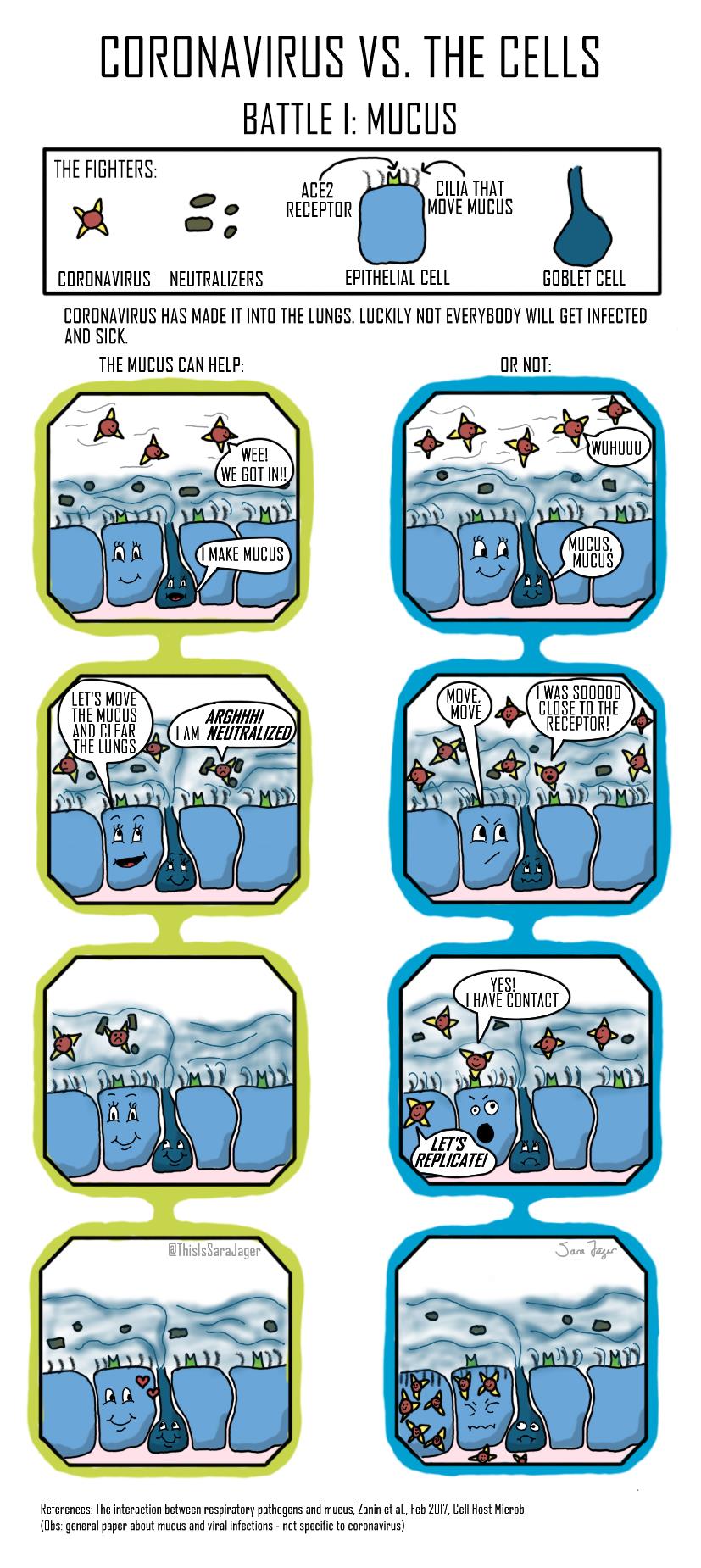
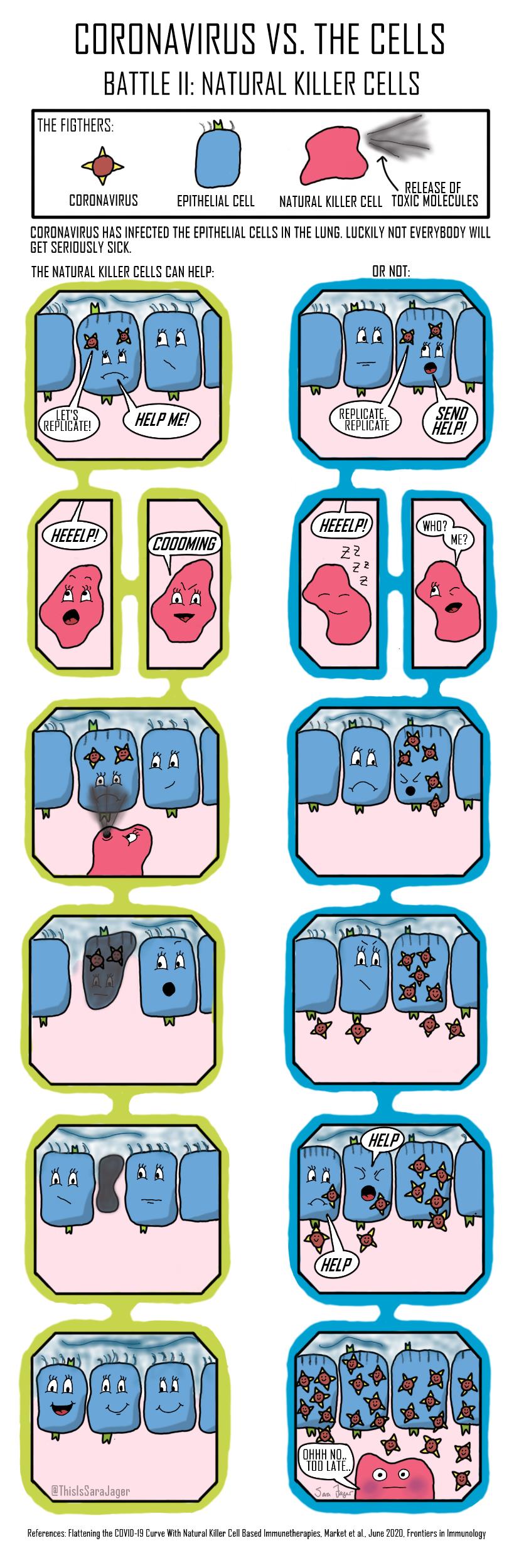
artwork by Sara Jager
Mahek Kothari
Undergrad at Dr Vikram Sarabhai Institute of Cell and Molecular Biology, the Maharaja Sayajirao University of Baroda.
How SARS-CoV-2 Triggers PANIC ATTACK
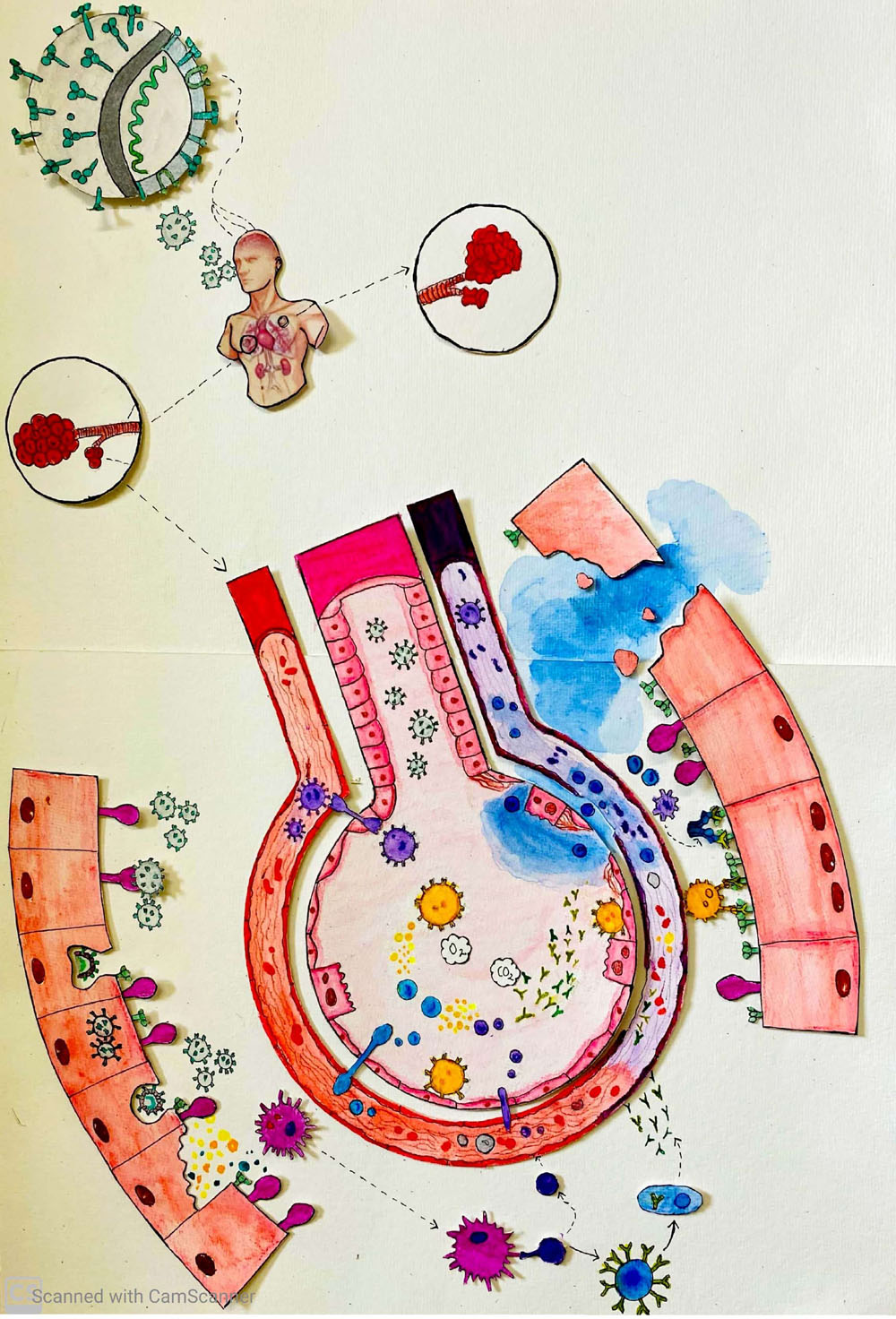
This illustration shows how the severe acute respiratory syndrome coronavirus (SARS-CoV-2) triggers “Prolific Activation of a Network-Immune-Inflammatory Crisis”, or “PANIC” Attack, in the alveolar tree of the lungs. The middle part illustrates the indivisible functional unit for gas-exchange, comprised of a single cell thick lining of epithelium consisting type I pneumocytes responsible for gas exchange; the type II pneumocyte secretes surfactant, crucial for reducing alveolar surface tension, which prevents alveolar collapse. Next I illustrate the aerosolized entry of SARS-CoV-2, which causes the accumulation of macrophages, mast cells, (PMNs), along with the cellular release of each cell’s effector elements, such as cytokines, chemokines, free radicals and ROS. On the outer part I have magnified the alveolar epithelium in order to illustrate the distinctive mechanisms which collectively represent the SARS- CoV-2 triggered PANIC. The release of new virions thereby perpetuates an amplification of the viral lifecycle and accelerates the destruction of lung alveoli, until a threshold burden of disease is established, beyond which the body’s continuous demand for oxygen can no longer be achieved, thus lungs collapse occurs and ultimately death of the patient.
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Mahek Kothari ”.
For more information contact me via: www.twitter.com/Mahek_3012
Christoph Kuehne
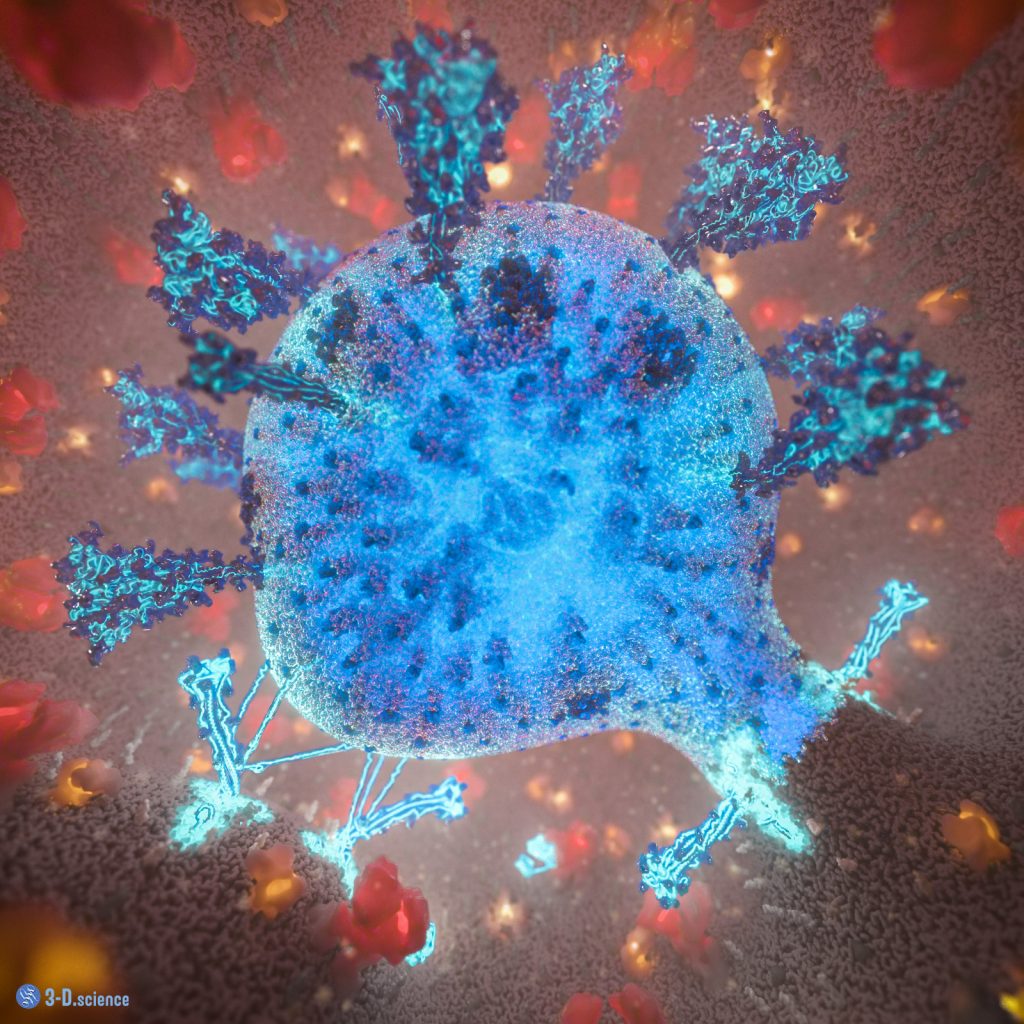
SARS-CoV-2 Fusion
When SARS-CoV-2 infects a cell, it docks to a receptor on its surface. The virus enters the cell enveloped in a portion of the cell membrane, forming a vesicle. The viral spike proteins then undergo a major transformation to fuse the membrane of the virus with that of its host. This image provides a glimpse into this process. The membrane fusion is already complete (lower right) while a second one is in progress (lower left). The viral genome (blue strands) enters the host cell to hijack its molecular machinery to produce more virus particles. Proteins of the cell are depicted in red (ACE2-BOAT receptor complex) and orange hues (TMPRSS2, ATPase, LAMP, Mucolipin, and Lipopolysaccharides).
License: This image is published under a CC-BY-NC 3.0 license. For use of this image, please acknowledge “Christoph Kuehne/3-D.science”.
Martina Maritan
Postdoc at the Center for Computational Structural Biology, Scripps Research, California
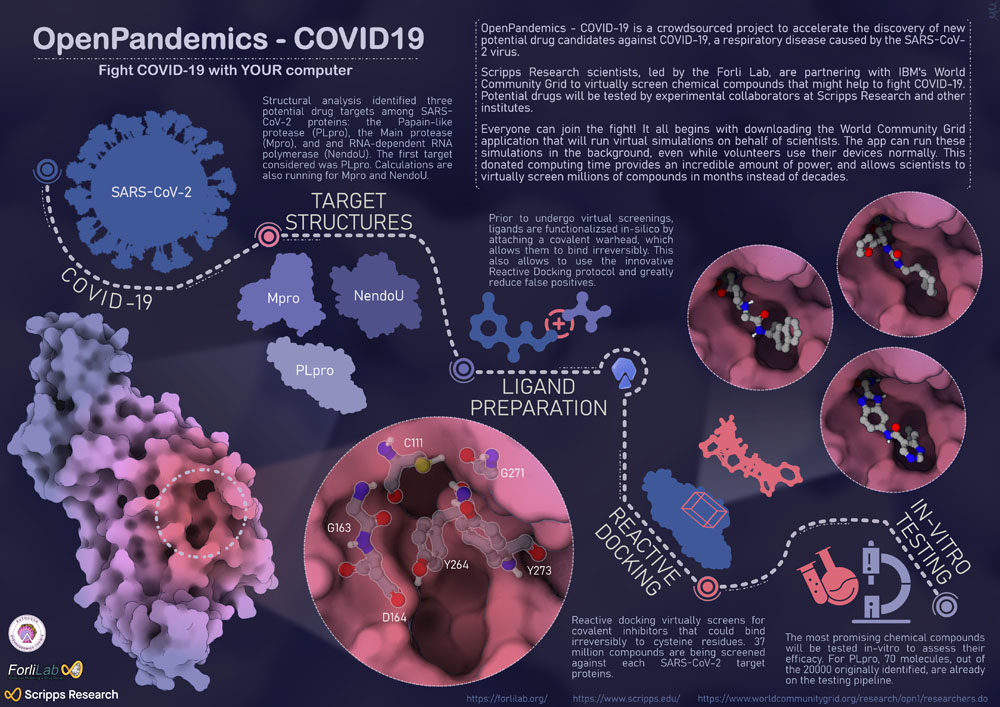
OpenPandemics – COVID-19
This infographic depicts the initial stage of the OpenPandemic-COVID-19 project and the early results achieved by Forli Lab at Scripps Research.
How can you help to fight COVID-19? Donate your unused computing power to scientists at Scripps Research to speed the discovery of potential drugs for the treatment of COVID. OpenPandemics-COVID-19 is a crowdsourced project in collaboration between IBM and Scripps Research, where anyone with a computer and an internet connection can help scientists to find compounds that might be effective against COVID-19. Volunteers just need to download the World Community Grid app: the software detects when the device is underutilized and available to work and directs unused computing power to run virtual experiments on behalf of scientists.
How does OpenPandermics-COVID-19 work? Researchers in Forli Lab started by focusing on three key SARS-CoV-2 protein targets: PLpro, Mpro and NendoU. Target structures have been analyzed to find the most promising druggable pockets. Millions of chemical compounds are virtually screened against these pockets and the best candidates will be experimentally tested in-vitro by experimental collaborators. The first viral target was PLpro. The initial screenings resulted in over 20000 compounds potentially binding PLpro. Further filtering and analysis brought down the number to 70 molecules, which will be tested in-vitro in the upcoming months. In parallel, millions of molecules are being docked onto Mpro and NendoU proteins and scientists are getting ready to test new viral targets. To know more visit https://www.worldcommunitygrid.org/research/opn1/overview.do.
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Martina Maritan.”
Mol Mir
Visual Artist and Laboratory Assistant, Stowers Institute for Medical Research
molmir.com
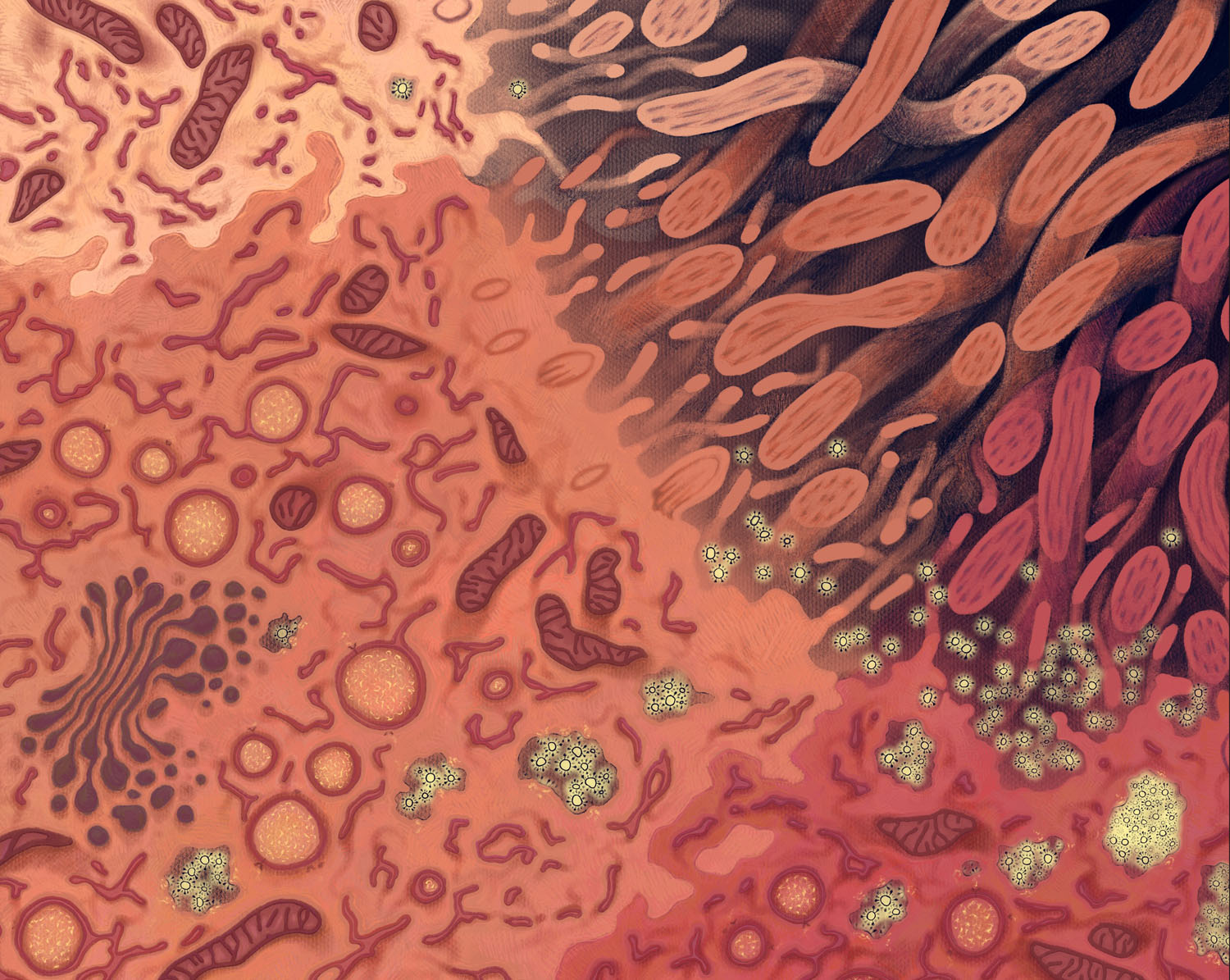
Infection: Illuminated
This artwork depicts three respiratory epithelial cells progressing through SARS-CoV-2 infection. Visible in the middle and bottom cells, endoplasmic reticulum has been transformed into a replication organelle, featuring double-membrane vesicles where viral RNA synthesis occurs. A close look reveals a molecular pore spanning this double membrane, releasing the RNA so that it can be encapsidated, travel to a virus assembly site, and bud into an endosome as a new virus. The pink cell in the bottom corner is releasing many new viruses, allowing infection to spread to new cells, tissues, and people.
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge Mol Mir.
For more information, you can find me here:
Email: mmir@stowers.org
Twitter: @molecularmir
Instagram: @molecularmirror
Website: molmir.com
Click here for a high-resolution file.
Sarina Mitchel

These paintings are based on microscope images of epithelial cells in human lungs. Epithelial cells line every surface of your body. These brave little cells form an essential barrier, separating one organ from another, outside from inside, you from the world. They let the good things like oxygen and nutrients through and keep the bad stuff out. When airway epithelial cells cannot perform their function as a barrier, a person can become sick with respiratory diseases like emphysema or COPD. They are also the main cells in your lungs that COVID-19 will attack if it gets too far, and this is where my art links in to the CellSpace 2020 workshop.
There is a huge gap between what most of us understand about our cells and bodies, and the research being done by scientists. The importance of science being more accessible has been made even clearer with recent events. With these paintings, I aim to turn the complexity of groups of airway epithelial cells into something beautiful, something that will intrigue viewers and inspire them to learn and ask questions.
My artistic process involves hand-tracing the cell boundaries, then programming a CNC router to etch that image into masonite (a dense, smooth board made from compressed wood fibers). As I paint, I try to find the right places to punch up that detail and specificity, but to still allow those beautiful moments of instinct, of searching, to shine. I often use bright gold to highlight the grooved outlines of the cells, coupled with iridescent inks to create a sense of depth and motion. I use these techniques together to create these shimmering, dimensional pieces of art.
Special thanks to Dr. Jennifer Mitchel at the Harvard School of Public Health for allowing me to use her microscope images and data.
License: please contact Sarina Mitchel via www.sarinamitchel.com for information on usage and permissions.
Instagram: @rina_the_beans
“Cells, Jammed”

“Moving in Concert”
“Cells, Unjammed”
Catarina Moreno
Illustrator & Graphic Designer, www.catarinamoreno.com
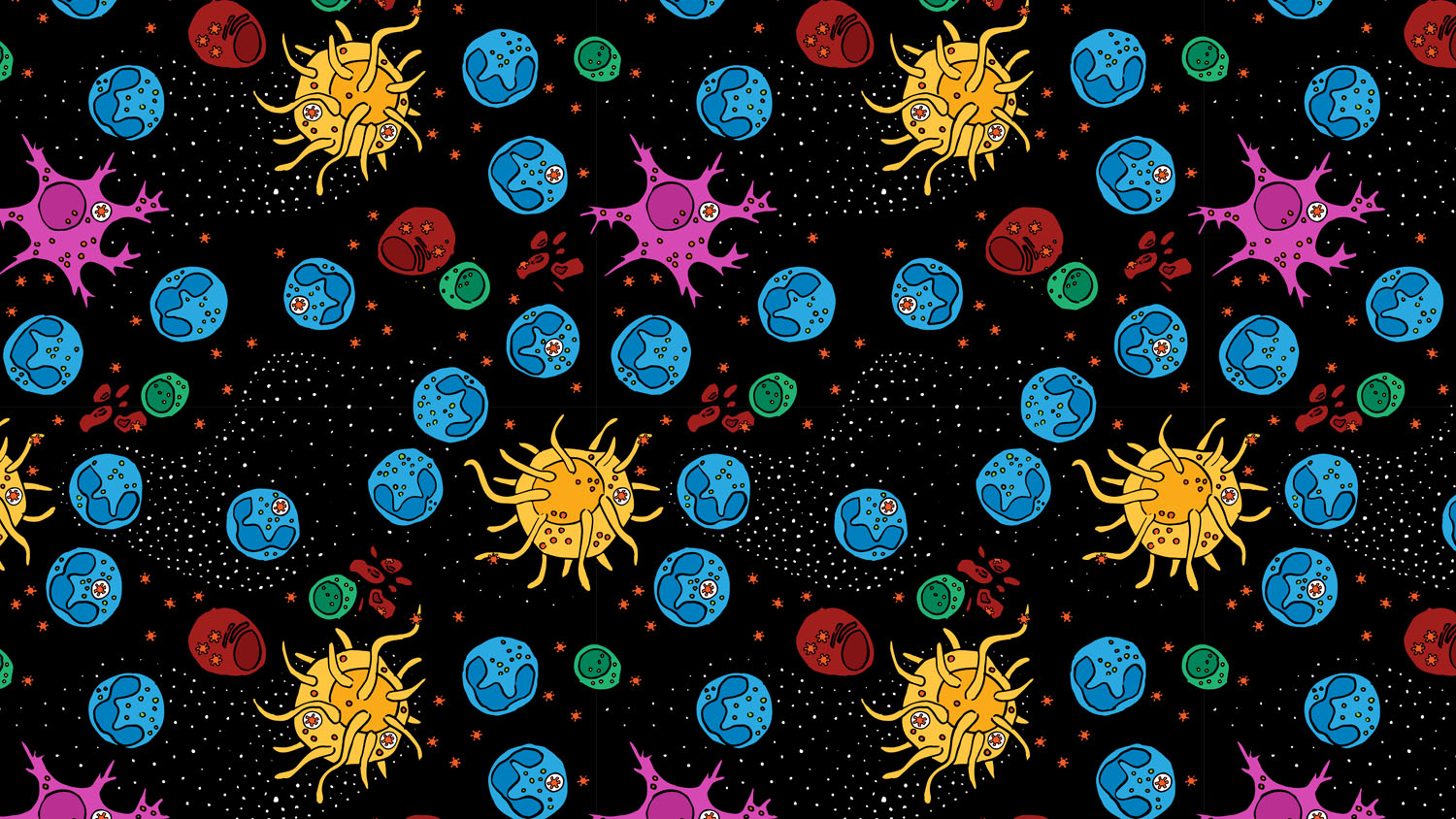
Innate Immune System (pattern)
Representation of the innate immune system fighting and winning against coronavirus. Wanted to a more kind of organic representation then usual but simple to identify the elements, based on microscope photos and illustrations, representing neutrophils, macrofages, dendritic cells, and NK cells.
Click here to download the pattern pdf file, which may be used as a tile to create a repeated pattern.
License: This work is provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Catarina Moreno”.
Shruti Morjaria
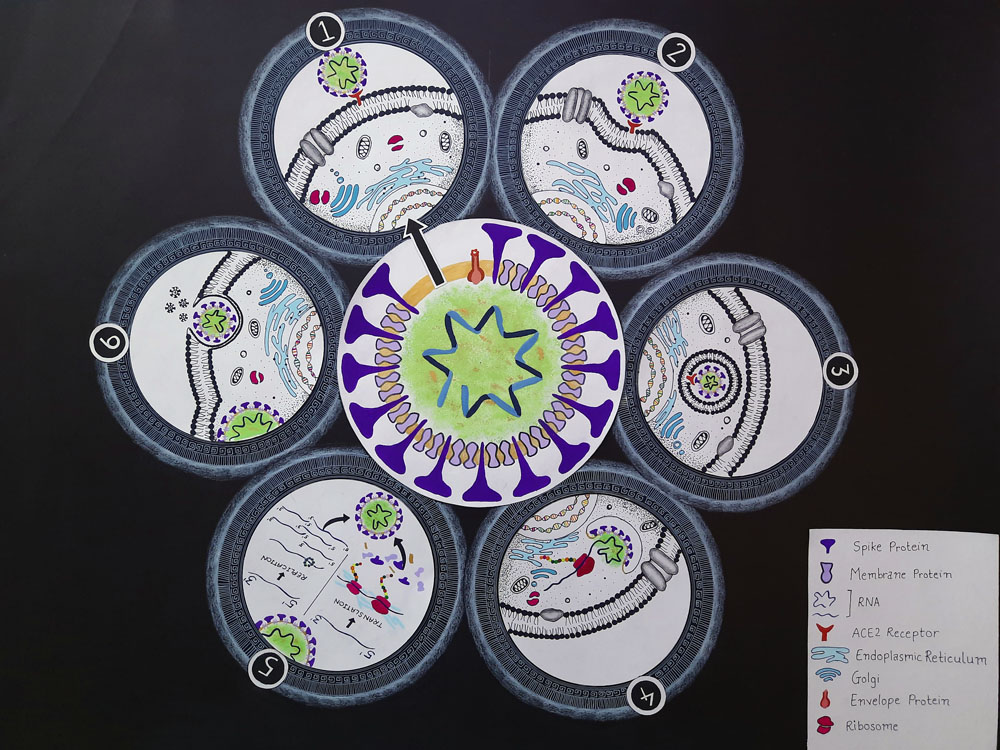
Coronavirus Replicative Cycle
This figure represents the replication of coronavirus : Once entered the host, the virus binds to the ACE2 receptor present on the cell surface (1). On binding the receptor, it is readily endocytosed into the cell (2) & (3) and the virus releases its genetic material into the cell (4). The virus has no machinery of its own for replication of its genetic material and translation of the same hence, it uses the host machinery to make more of its proteins and genetic material. Once the genetic material and protein are made, they assemble to form new copies of the virus (5). The new copies of the virus are exocytosed and further released which infect other cells and survive in the host (6).
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Shruti Morjaria”.
Arthur J. Olson
How Small is the SARS2 Coronavirus?
The size of a virus particle is unimaginably small, and is actually unseeable by the human eye since it is smaller than the wavelength of visible light. We can best get a sense of its size by showing it in the context of other objects that range from familiar things that we can see, down to those that we cannot see unless aided by technology. I created a movie and an interactive experience that attempts to do this, with relatively simple representations, but with realistic relative dimensions. The movie takes the viewer linearly from the familiar world of small objects to the microscopic world and beyond. The interactive visualization allows the viewer to explore in a more free-form manner these size relationships. This can most dramatically be experienced in a virtual reality where viewers can feel like they have been shrunk down to the size of their microscopic surroundings.
One significant point is made at the end of the movie. The virus particle, or virion, is actually not the virus itself. Since a virus needs a host and a means of transmission to complete its continuity. Thus, the tiny virion is only one stage in the life cycle of the virus, and as we have sadly come to see, a successful virus like the CoV-SARS2 can in fact attain the size of the entire globe.
Isha Parlikar
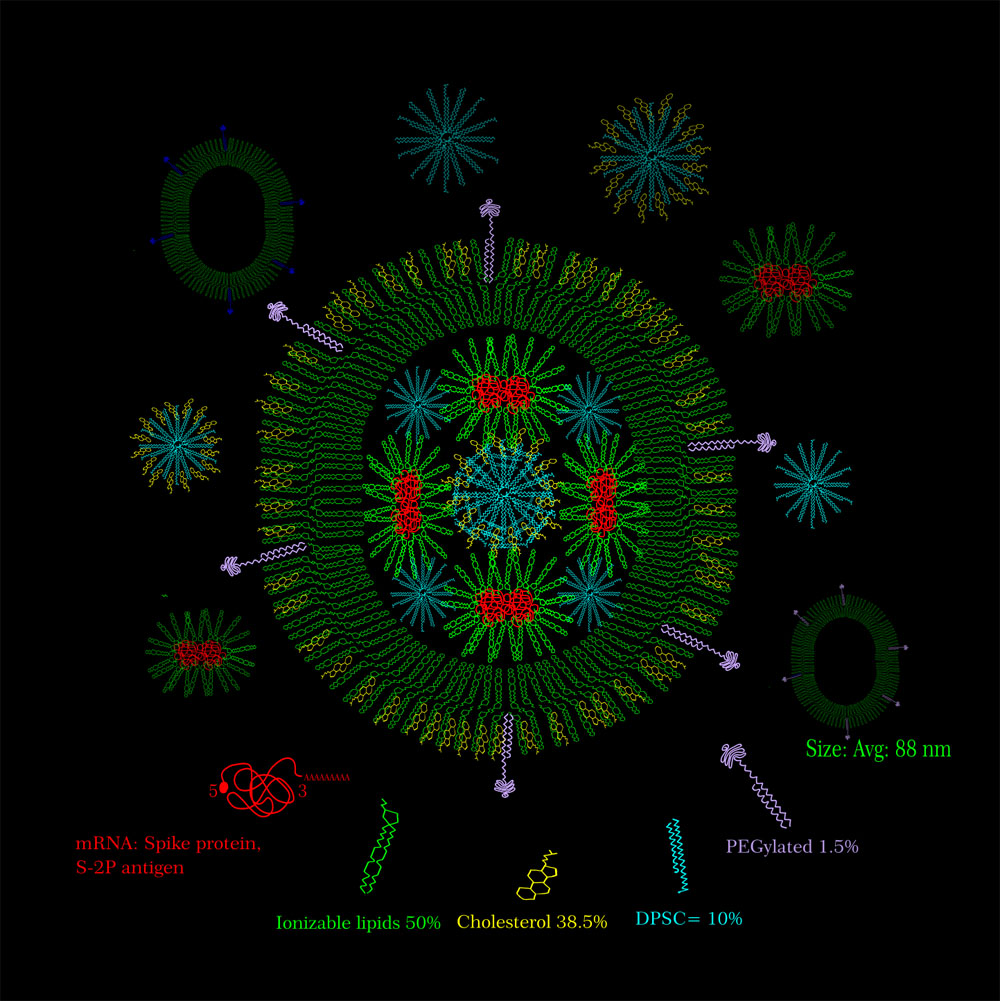
Moderna Vaccine Candidate mRNA1273
Vaccine candidate of Moderna mRNA-1273, is a S-2p antigen of SARS-CoV-2 spike protein that is stabilized in perfusion conformation. The artificially synthesized mRNA is encapsulated into liquid nanoparticles (LNPs) containing ionizable lipids. The LNPs are in 50:10:38.5:1.5 ratio (ionizable lipid:DSPC:cholesterol:PEG lipid).
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Isha Parlikar”.
Spike Protein Dance
A collaboration with Devika Nair
This video explores the flexibility of spike protein on coronavirus with the help of Indian classical dance Kathak. We hope that by sharing the story and the events happening around the spike protein with the help of traditional dance, we can help make people aware and inspire for their safety.
Special Thanks to my Guru Shrimati Prerana Deshpande, Founder at Nrutyadham, Pune
License: Please contact isha@parlikar.com for information on usage and permissions
Onisha Patel
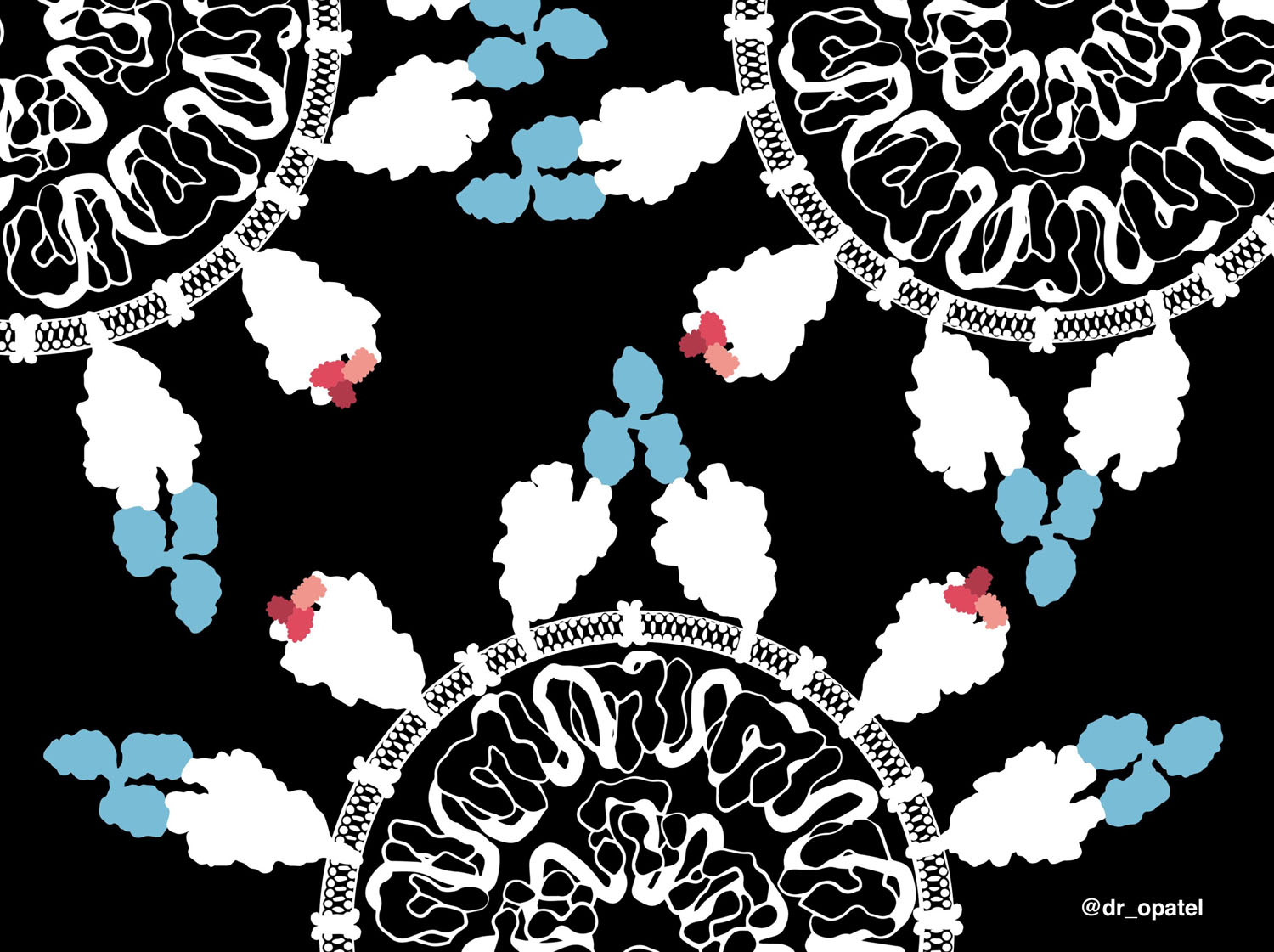
Minibinders, small antiviral proteins for SARS-CoV-2 therapeutics
This artwork shows a comparison between minibinders (small antiviral proteins, shown in shades of red) and antibodies (blue) that bind to the spike proteins on the surface of the SARS-CoV-2 virus. Prof David Baker’s group at the Institute of Protein Design commenced the computational design of minibinders in January 2020 around the time when community transmission of the virus began. By the end of May, they had identified minibinders that were able to strongly bind the spike proteins and neutralize the virus. Minibinders are 20-fold smaller than antibodies, and can be scaled up in production at a lower cost. While antibodies have only two binding sites to its target, minibinders in an equal mass offer 20-fold more potential neutralizing sites. The small size and higher stability of minibinders offer a possible advantage of nasal formulations for drug delivery and eliminating the need for refrigeration during storage and transport. These minibinders are currently in development for their potential use as SARS-CoV-2 therapeutics. This example highlights the power of computer-generated protein design, especially during a pandemic when time is critical.For more information refer to https://www.bakerlab.org/index.php/2020/09/09/covid-minibinders/
Thank you to Longxing Cao and David Baker for providing the PDB coordinates of minibinders bound to the spike protein before the actual release date.
Twitter and Instagram: @dr_opatel
Licence: This work is freely available with a CC-BY-4.0 license. For use of this image, please acknowledge “Onisha Patel”.
Reference: Cao, L et al. (2020) De novo design of picomolar SARS-CoV-2 miniprotein inhibitors. Science, 370 (6515), 426-431. DOI: 10.1126/science.abd9909
Kadambari Patil
Dizziness
A lot of studies recently have revealed dizziness as one of the most common neurological manifestation of COVID-19. Thus it should not be taken lightly and we should remain vigilant. Here’s a dizzy effect on the COVID-19 to emphasize not ignoring any symptoms! Vestibular rehabilitation therapy has shown promising results against dizziness in stable COVID-19 patients.
License: These media are provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Kadambari Patil.”
For more information contact @kad_leidoscope (Instagram), writetokadambari7900@gmail.com

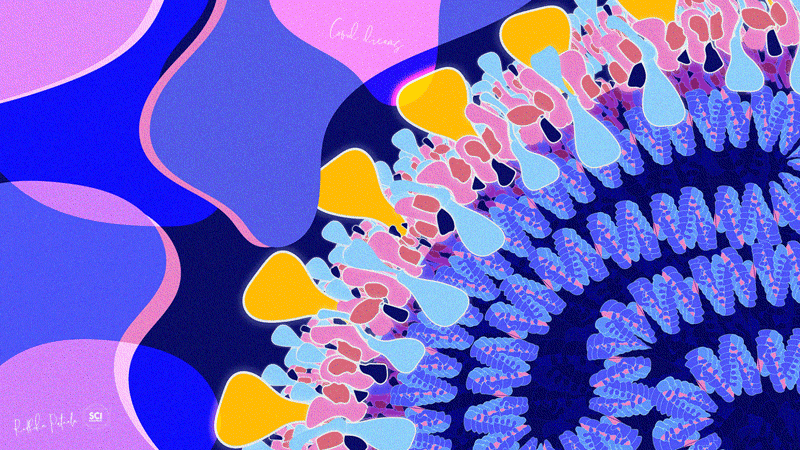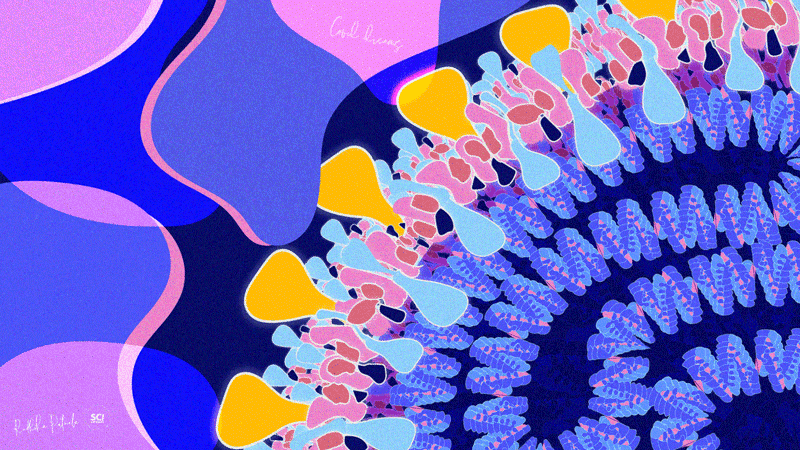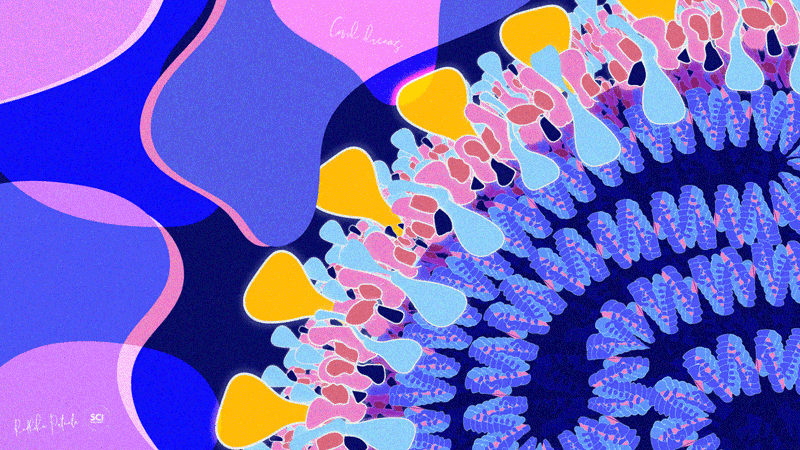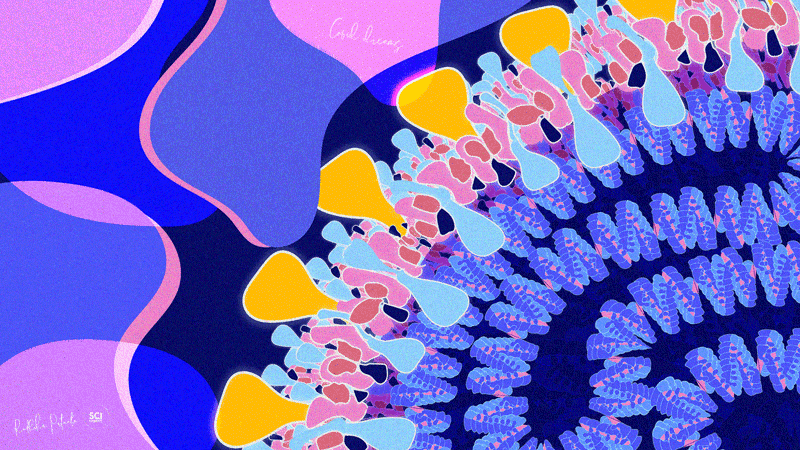
Dr. Radhika Patnala
Covid Dreams
A lil’ something to get by these quarantine days, “Covid Dreams” are a collection of abstract art in mixed media that capture some mixed feelings related to the pandemic.
License: For permission to use please contact radhika@sci-illustrate.com.
Visit the Sci-Illustrate site to learn more about the artwork.



Himasha Mariann Perera
Doctoral Student, Department of Chemistry & Biochemistry, Baylor University, Waco, Texas
Mask Up Your Lungs: Help Prevent Spreading of the Virus
Mask Up Your Lungs depicts the importance of wearing a mask to protect your lungs from SARS-CoV-2. Wearing a mask have become the new normal and it will continue to be for a while. In an abstract artistic approach, this creation depicts the difference between an infected lung (left) attacked by the virus and a healthy lung protected by masks (right). The artistic impression behind this creation is that a respiratory droplet carrying the virus enters the respiratory system, but fails to attack the Masked Up Lung. The two assemblies of alveoli in the infected lung emphasize the severe and moderate impacts of SARS-CoV-2 and the subsequent immune response.
License: These media are provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Himasha Mariann Perera”. For more information contact jkhimashaperera@gmail.com or @sci_art_by_h (Instagram).

Hailee Perrett
Coronavirus anatomy explained
This video introduces coronaviruses, a large family of viruses, some of which infect humans.The coronavirus at the root of COVID-19 is the newest known member of this family, and like other coronaviruses that infect people, the new coronavirus causes respiratory disease, among other symptoms.
How the novel coronavirus infects a cell
Due to its unique features, the novel coronavirus is particularly good at infecting new cells, both in the upper respiratory tract, as well as deeper down in the lungs. Here’s a look at how the process takes place.
How antibodies neutralize the novel coronavirus
An animated look at the immune response to the novel coronavirus that causes COVID-19. Learn how scientists are studying the antibodies produced by people exposed to SARS-CoV-2. These antibodies could be used to create treatments or vaccines for the virus.
These videos were created in the laboratory of Andrew Ward at the Scripps Research Institute, in association with CellSpace and the TSRI SciComm team.
License: This work is freely available with a CC-BY-4.0 license. For use of this video, please acknowledge “Drew Duglan, Faith Hark, Hailee Perrett, and Matthew O’Connor.”
Manasi Pethe
Pethdoodles
Life in the times of COVID
You are no stranger to the virus – COVID-19. This is a virus that affects the upper and lower respiratory tracts and most affects the lungs along with other adverse side effects. The best way to protect yourself and others from this is to maintain distance, wear a mask!, and wash hands frequently along with other measures especially reducing interactions with other households. COVID -19 has changed how we interact with one another. With widespread differences in questioning whether the virus even exists(yes, it does!!) to asking – well can 200 people be in my social bubble? I decided to make a social bubble mandala to portray the same.
License: This image provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Manasi Pethe, PethDoodles (C) 2020”
Click here for a high resolution version of this artwork.
Find more of Manasi’s work on Instagram, Facebook and Twitter.

The mandala portrays a story – a person who wanders out of his trusted bubble to go meet a friend for her birthday. This birthday party – a crowded, unsafe and potentially super spreader event – further branches into three scenarios –
- First, the very same person who tells his trusted circle that he thinks he should isolate since he may have been exposed.
- Second, a person blissfully oblivious to the existence of the virus goes out further to a nightclub and doesn’t bother to even wear a mask – a potentially fatal action for millions
- Third, a person so ridden with anxiety around the virus, they have barely met a single person since the pandemic began.
Which one of these people do you relate to the most? The pandemic faces us with multiple important choices on a daily basis and it is important to continue to choose for safety, wear a mask PROPERLY (over your nose and mouth), for ones mental health and for the health of those around you – perhaps some people you may have never met!
#staysafe
Mrs. Revathi Ravishankar
independent researcher ~ bioartist ~ science educator ~ bioinformatician ~ microbiologist
MSc Microbial Biotechnology
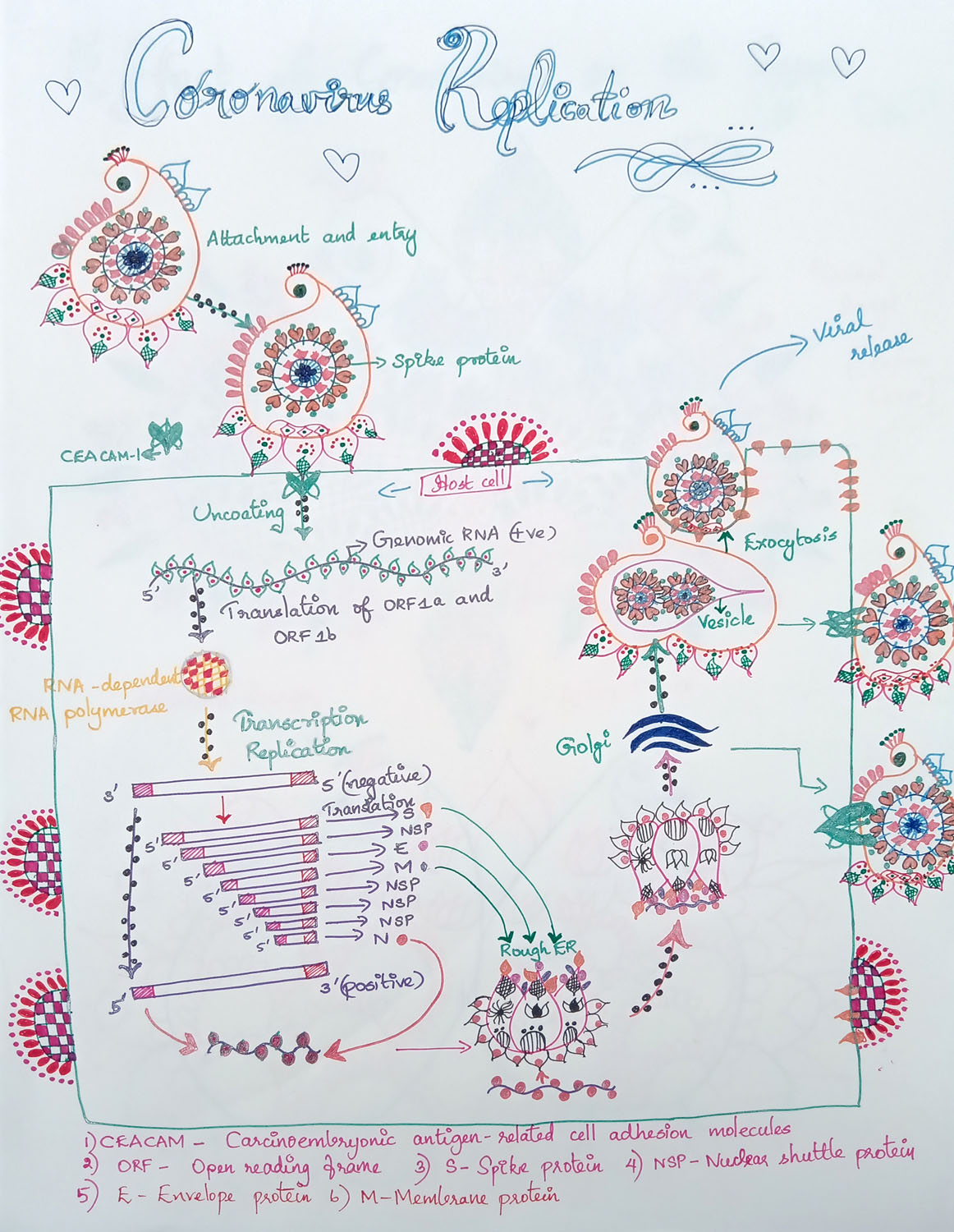
Coronavirus Replication

Effect of Coronavirus on the Lungs
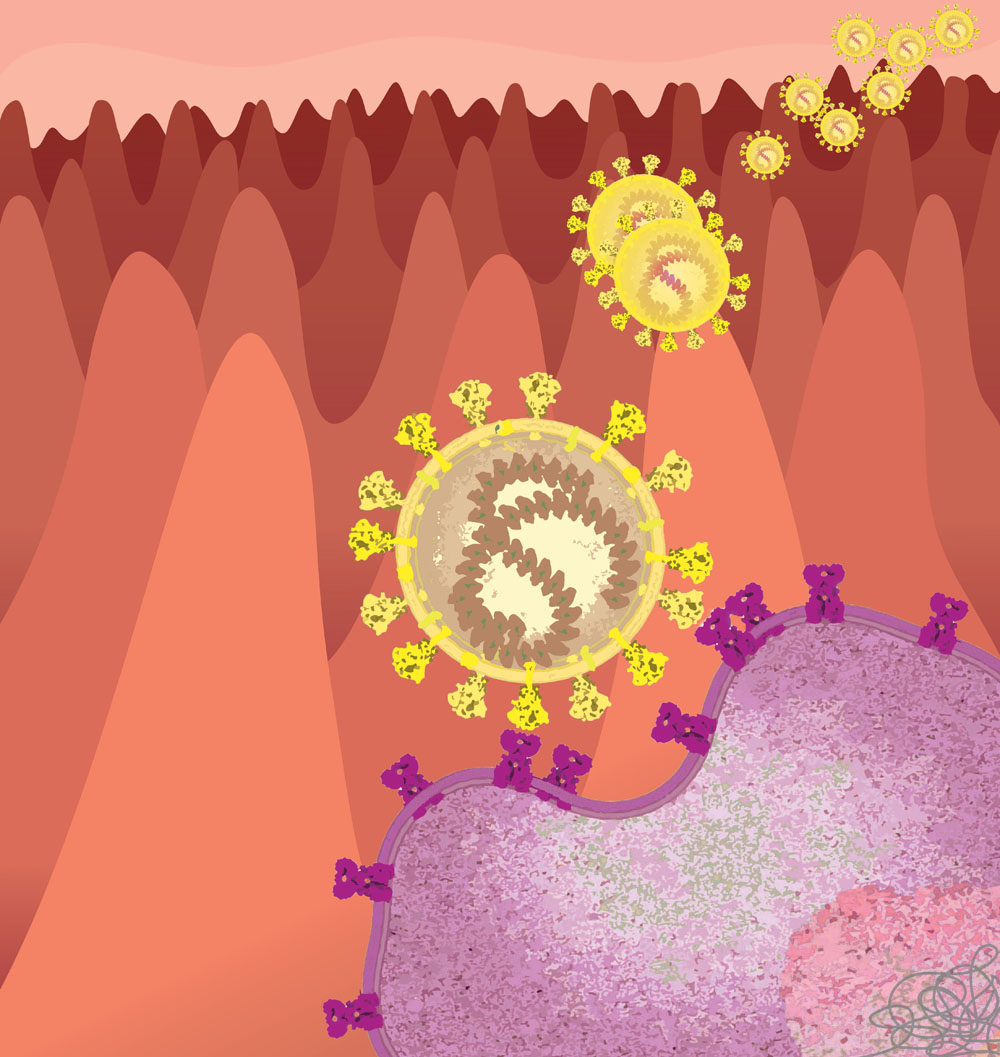
Malavika Saji
SARS-CoV-2 Entry
The picture shows how the SARS-CoV-2 enters the cell. It uses the viral spike protein (S-protein) to bind to the human ACE-2 receptor (hACE-2) through its receptor binding domain (RBD) and undergoes proteolytic activation by human proteases. The high affinity of hACE-2 with the RBD makes the viral entry more efficient while evading recognition by the immune system. This is one of the major reasons for the wide spread of the disease.
License: This media is provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Malavika Saji”.
Click here for a high-resolution file.
For more information contact:
E-mail: malavikasaji7@gmail.com
Twitter: @MalavikaSaji07
Instagram: _.lub.dub

Shehryar (Shay) Saharan and Naomi Robson
An ss-design (www.ss-design.site) and Robson Visuals (www.robsonvisuals.com) collaboration

What We Know So Far: COVID & The Brain
Many patients of all ages have been reporting brain fog and intense exhaustion months after recovering from even mild COVID-19. These brain-related complications of COVID-19 are seen throughout the course of the disease. What are they? Why are they happening? Follow the QR code to find out!
To see our full infographic: https://tinyurl.com/y52k7w8u
Contact: s.saharan@mail.utoronto.ca @Shay_Sah (Twitter), @shehryar.art (Instagram); naomi.robson@mail.utoronto.ca, @robson_visuals (Twitter and Instagram)
License: This work is freely available with a CC-BY-4.0 license. For use of this image, please acknowledge Shehryar (Shay) Saharan, and Naomi Robson.
Shivam Sharma
A Gut Feeling
Sars-CoV-2 is known to infect the lungs transmitting primarily through respiratory air drops. However, there are evidences that prove that the intestine can also be another viral target organ as the receptor Angiotensin- converting enzyme 2 (ACE 2) is highly expressed in differentiated enterocytes. The artwork depicts the binding of Spike protein on Sars-CoV-2 with the ACE 2 receptor on enterocyte cell. This suggests that Sars-CoV-2 may also be an enteric virus and transmit though a secondary i.e. faecal-oral route.
License: This image is provided for free use under a CC-BY-4.0 license. For the use of this image, please acknowledge “Shivam Sharma.”
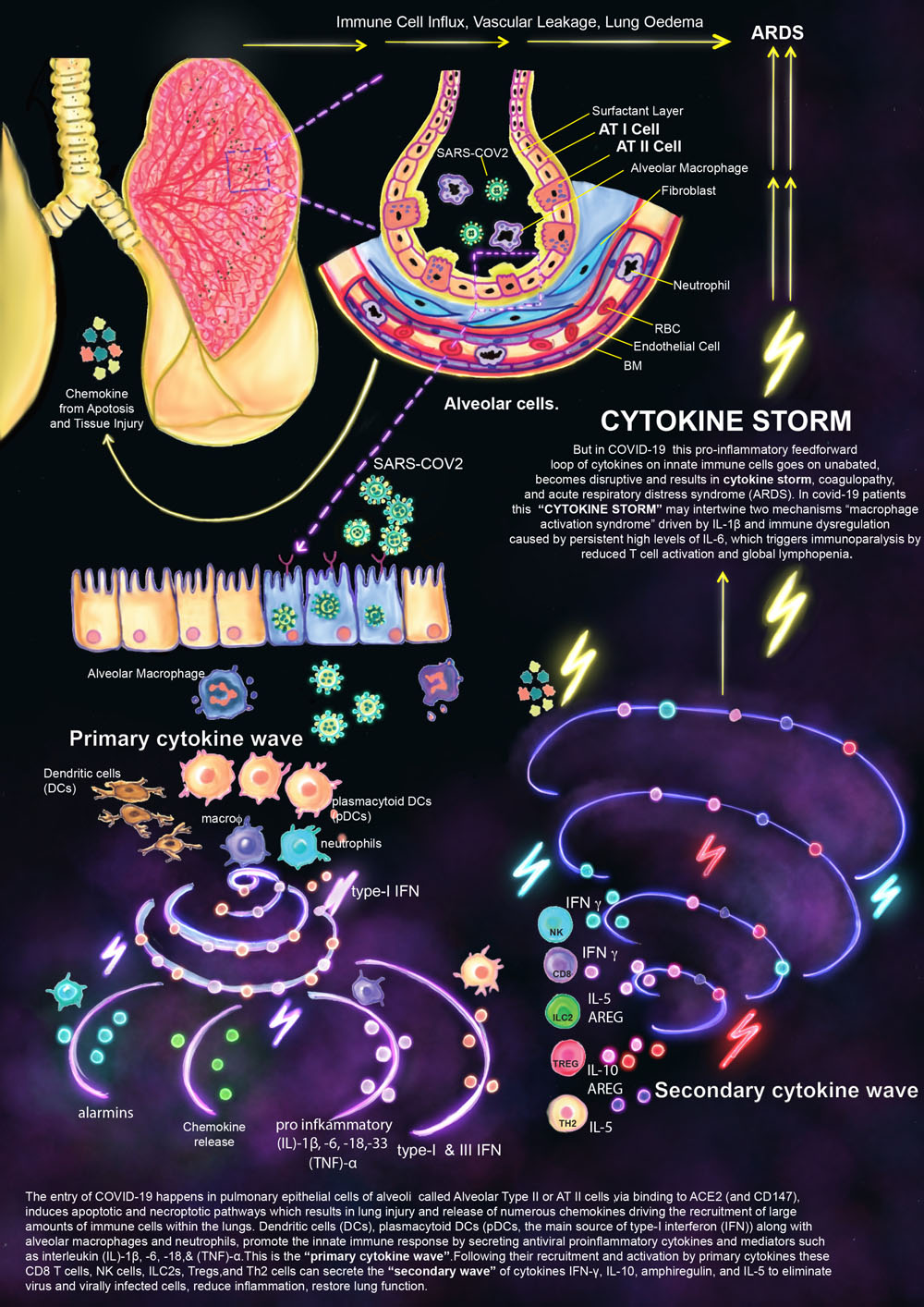
Dr. Shashikala Sasidharan
Postdoctoral student at RWJMS, Rutgers University
Cytokine Storm
The entry of COVID-19 happens in pulmonary epithelial cells of alveoli called Alveolar Type II or AT II cells, via binding to ACE2 (and CD147), induces apoptotic and necroptotic pathways which results in lung injury and release of numerous chemokines driving the recruitment of large amounts of immune cells within the lungs. Dendritic cells (DCs), plasmacytoid DCs (pDCs, the main source of type-I interferon (IFN)) along with alveolar macrophages and neutrophils, promote the innate immune response by secreting antiviral proinflammatory cytokines and mediators such as interleukin (IL)-1β, -6, -18, & (TNF)-α. This is the “primary cytokine wave.” Following their recruitment and activation by primary cytokines these CD8 T cells, NK cells, ILC2s, Tregs, and Th2 cells can secrete the “secondary wave” of cytokines IFN-γ, IL-10, amphiregulin, and IL-5 to eliminate virus and virally infected cells, reduce inflammation, restore lung function.
But in COVID-19 this pro-inflammatory feedforward loop of cytokines on innate immune cells goes on unabated, becomes disruptive and results in cytokine storm, coagulopathy, and acute respiratory distress syndrome (ARDS). In covid-19 patients this “CYTOKINE STORM” may intertwine two mechanisms “macrophage activation syndrome” driven by IL-1β and immune dysregulation caused by persistent high levels of IL-6, which triggers immunoparalysis by reduced T cell activation and global lymphopenia.
License: This image is provided for free use under a CC-BY-4.0 license. For the use of this image, please acknowledge “Shashikala Sasidharan.”
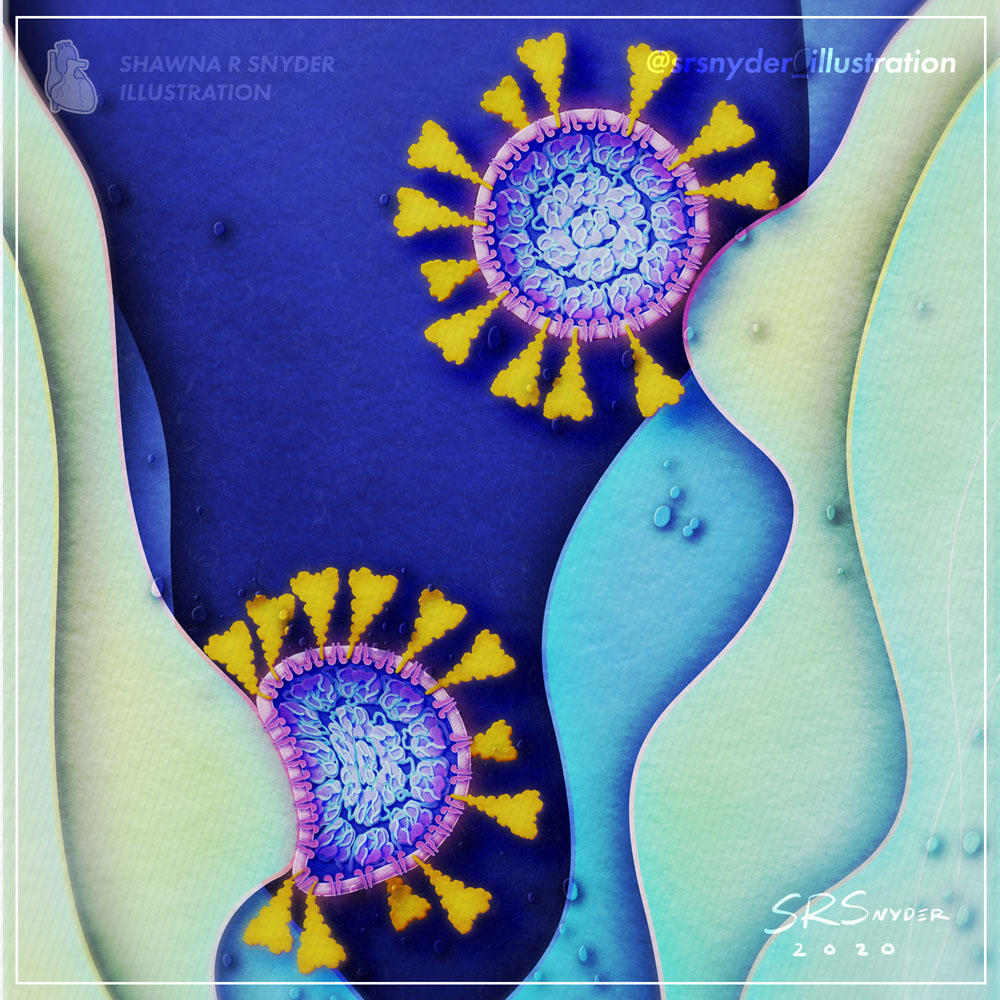
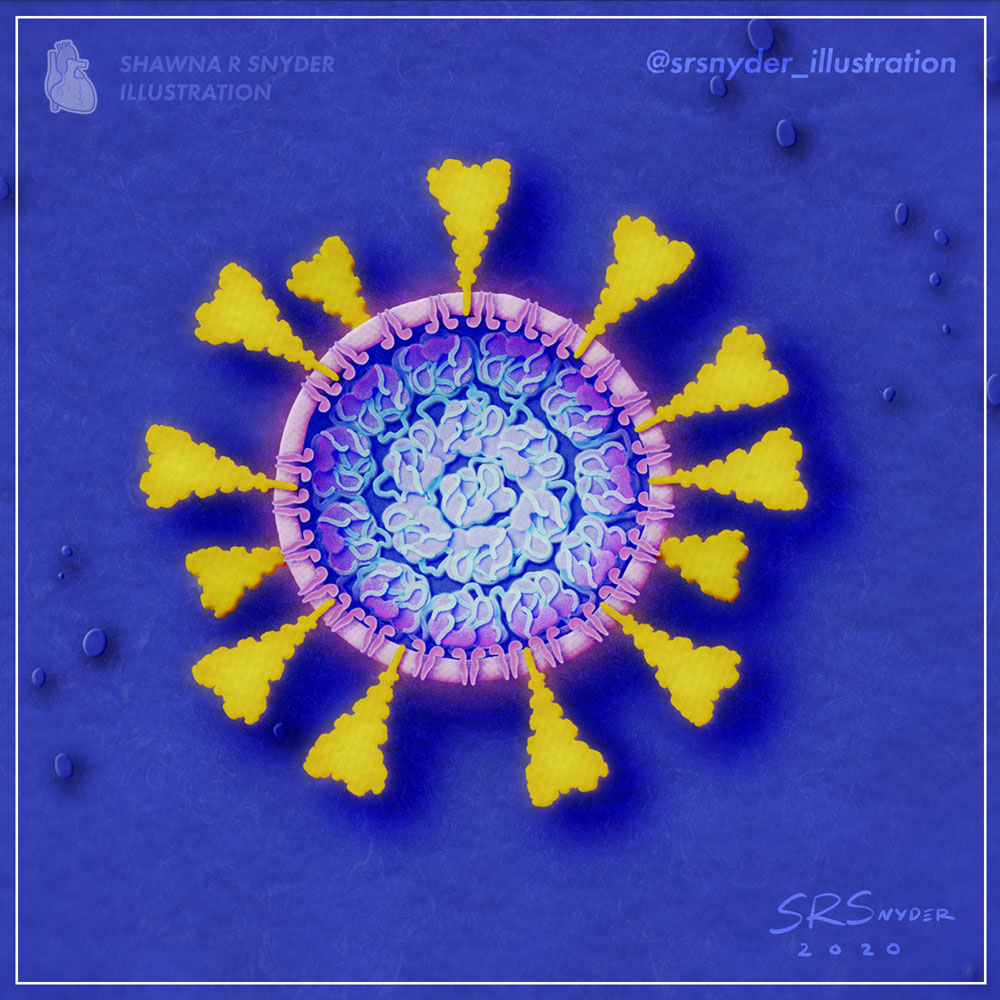
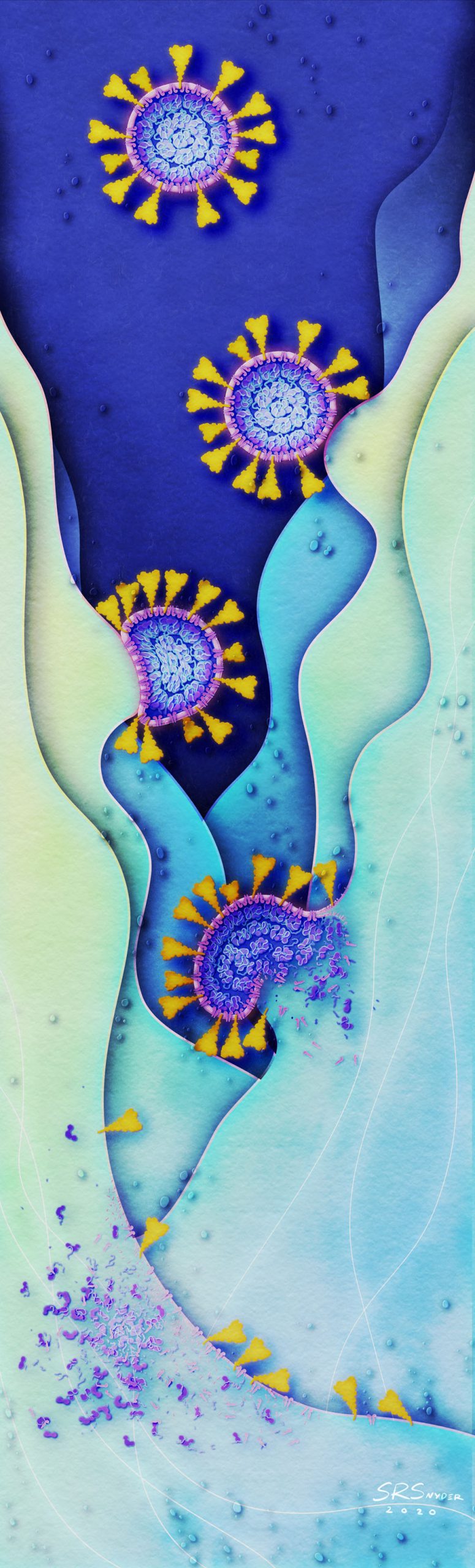
Shawna R Snyder, M.A.
Medical Animator & Visual Artist, www.srsnyderillustration.com
Dismantlement of the COVID-19 virus with Soap
As the pandemic rages, one of the simplest ways to protect yourself and prevent COVID-19 is to wash your hands with soap. Soap films consist of molecules that can penetrate and weaken the viral membrane. The combined soaping motion (rubbing your hands together) further disrupts and dismantles the virion. Remember: just 20-30 seconds of handwashing is all it takes to dismantle the virus that causes COVID-19.
License: Please contact Shawna Snyder for information on usage and permissions.

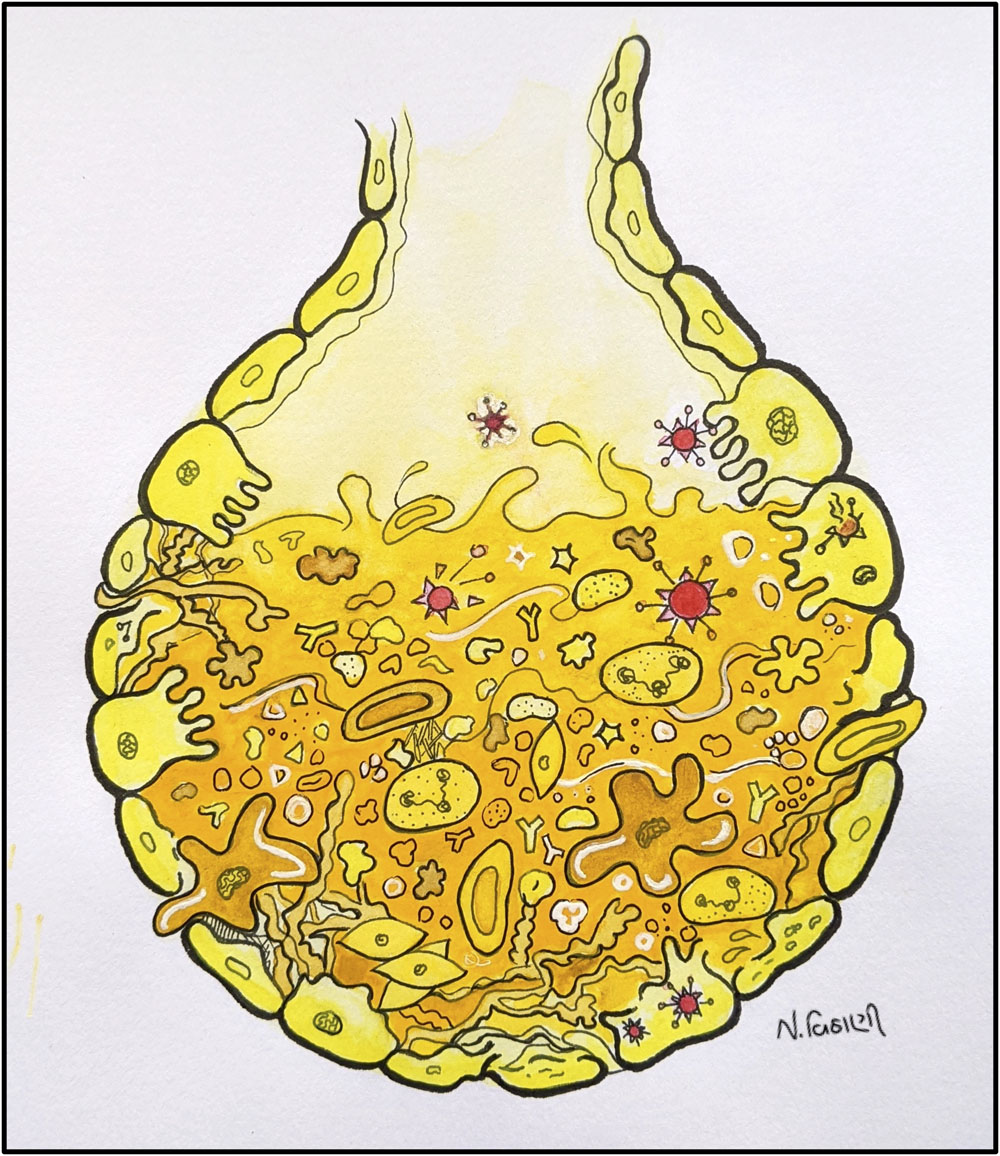
Neha Vithani
What coronavirus does to your lungs
Does this little sac look like a junkyard? That’s what coronavirus does to the sac-like structures called alveoli in the lungs. In a normal condition, the alveolus is filled with air for gas exchange. In severe coronavirus infection, it gets filled up with fluid & debris causing shortness of breath.
License: This work is provided for free use under a CC-BY-4.0 license. For use of this image, please acknowledge “Neha Vithani”.
Works in Progress
Mask Master Trivia!
You may already know how to wear a mask properly, but these trivia questions will show you if you’re really a Mask Master! Created by sci-artists Samantha Johnson, Gloria Fuentes, Neha Vithani, and Himasha Perera; this trivia video will really test your safety strategies in the age of COVID-19.